Integrating AI, Multi-Omics, and Nanomedicine in Cancer Therapy: A 5-Year Update across Major Solid Tumors
Keywords:
smart nanocarriers, multi-omics, precision oncology, liver cancer, breast cancer, renal cell carcinoma, glioblastoma, RNA therapeutics, immuno-nanotherapy, theranosticsAbstract
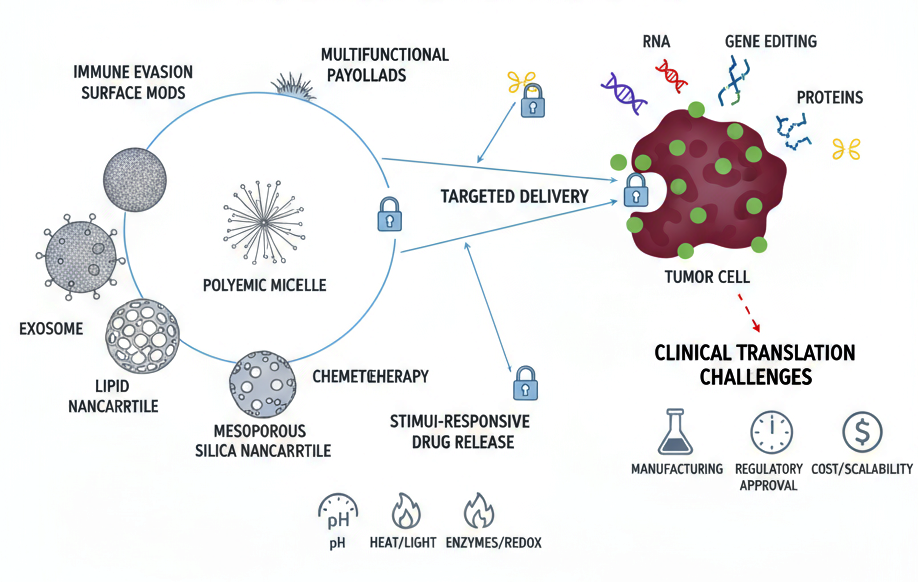
Background: Between 2019 and 2024, smart nanocarriers have emerged as a transformative platform in precision oncology, offering targeted, stimuli-responsive, and multifunctional delivery systems capable of addressing the limitations of conventional cancer therapies. Simultaneously, multi-omics technologies spanning genomics, transcriptomics, proteomics, and metabolomics have enabled patient-specific therapeutic design, allowing nanomedicines to be precisely matched to tumor molecular profiles. Objectives: This review synthesizes recent advances in nanotechnology-based approaches for liver, breast, kidney, and neuro (brain) cancers, highlighting how innovations in nanocarrier design, payload engineering, and tumor microenvironment modulation are being integrated with multi-omics insights to achieve personalized therapy. Methods: A comprehensive analysis of literature and clinical trial data from 2019–2024 was conducted, focusing on nanoparticle classes, payload strategies, surface functionalization, omics-guided targeting, and translational progress. Comparative assessments across the four cancer types were made to elucidate common challenges and cancer-specific solutions.Results: Smart nanocarriers have enabled RNA-based therapeutics, immuno-nanotherapies, photothermal and photodynamic strategies, and BBB-penetrating systems, demonstrating significant preclinical success and emerging clinical validation. Omics-guided design has refined patient selection, enhanced therapeutic efficacy, and facilitated the development of diagnostic and theranostic platforms. Key translational challenges include scalable manufacturing, regulatory compliance, long-term safety, and clinical validation.\ Conclusions: The integration of smart nanocarriers with multi-omics-guided personalization represents a paradigm shift in precision oncology. AI-driven nanoparticle optimization, combined with adaptive clinical trial designs, promises to accelerate clinical translation, positioning smart nanomedicine as a central component of individualized cancer therapy across liver, breast, kidney, and neuro cancers.
Downloads
Published
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.





