Nanotheranostics in Multi-Organ Cancer Management: From Liver to Brain Tumors via Omics and Artificial Intelligence
Keywords:
Nanotheranostics; AI; multi-omics; precision oncology; liver cancer; breast cancer; kidney cancer; brain tumors; smart nanocarriers; RNA therapeutics; immunotherapy; theranosticsAbstract
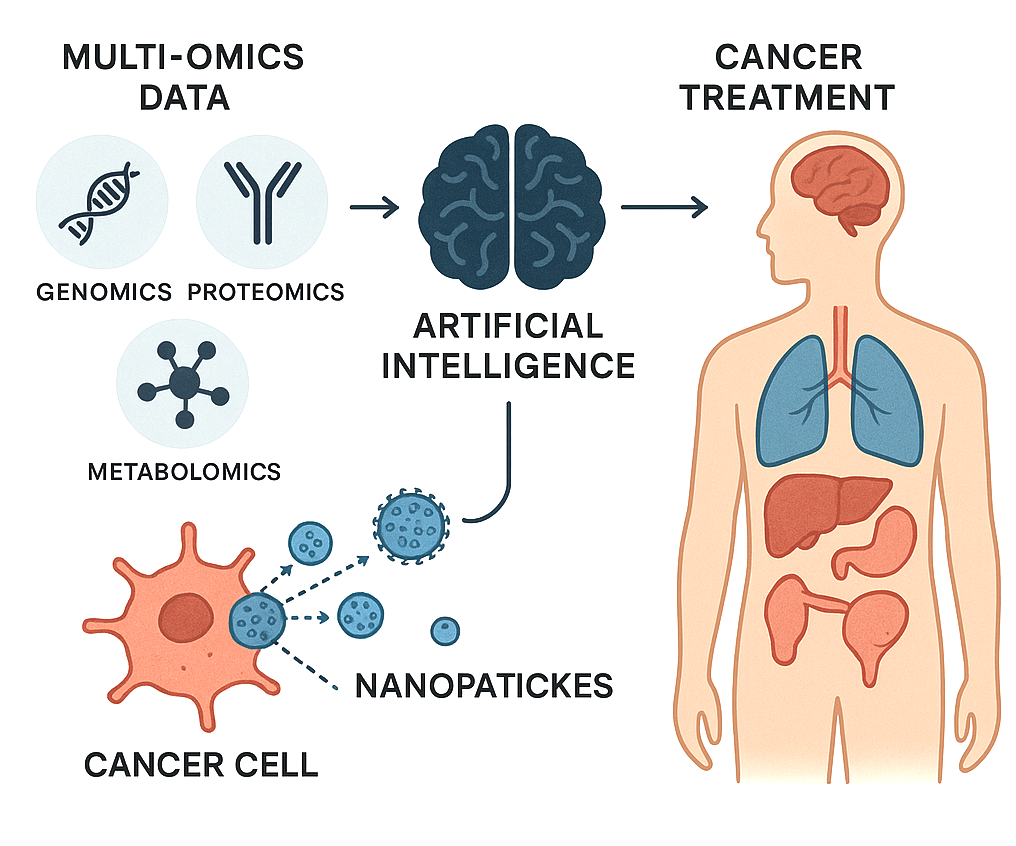
Background: From 2019 to 2024, nanotheranostics multifunctional nanosystems combining therapeutic and diagnostic capabilities have emerged as a transformative approach in oncology. The integration of artificial intelligence (AI) and multi-omics technologies enables precise tumor targeting, patient-specific therapy personalization, and real-time monitoring of treatment response across major solid tumors. Objectives: This review provides a comprehensive update on recent advances in nanotheranostics for liver, breast, kidney, and brain cancers, highlighting cancer-specific strategies, multi-omics-guided targeting, AI-assisted optimization, and translational progress. Methods: Literature from 2019–2024 was systematically evaluated, focusing on nanocarrier types, therapeutic and diagnostic payloads, functionalization strategies, and integration with AI and multi-omics. Cancer-specific applications, translational barriers, and regulatory considerations were analyzed comparatively. Results: Advances in liposomes, lipid nanoparticles, polymeric carriers, biomimetic nanoparticles, and exosomes have enabled multifunctional delivery of chemotherapeutics, RNA therapeutics, immunomodulators, and CRISPR-Cas systems. Diagnostic payloads, including MRI, CT, PET agents, and biosensors, allow real-time monitoring of therapeutic response. Multi-omics insights guide patient stratification and payload selection, while AI predicts nanoparticle behavior and optimizes design. Preclinical and early clinical studies demonstrate promising efficacy, although challenges remain in manufacturing, safety, and regulatory compliance. Conclusions: The convergence of nanotheranostics, AI, and multi-omics represents a paradigm shift in precision oncology. Future directions include AI-guided adaptive nanocarriers, omics-informed patient personalization, and adaptive clinical trial frameworks. Addressing translational and regulatory challenges will be key to realizing the full clinical potential of these integrated strategies, improving outcomes across liver, breast, kidney, and brain tumors.
Downloads
Published
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.





