Nanotechnology in Targeted Oncology: Progress Clinical Trials and AI-Omics Integration
Keywords:
Nanomedicine; Precision Oncology; Clinical Trials; Artificial Intelligence; Omics; Personalized Cancer Therapy; Drug Delivery; Theranostics.Abstract
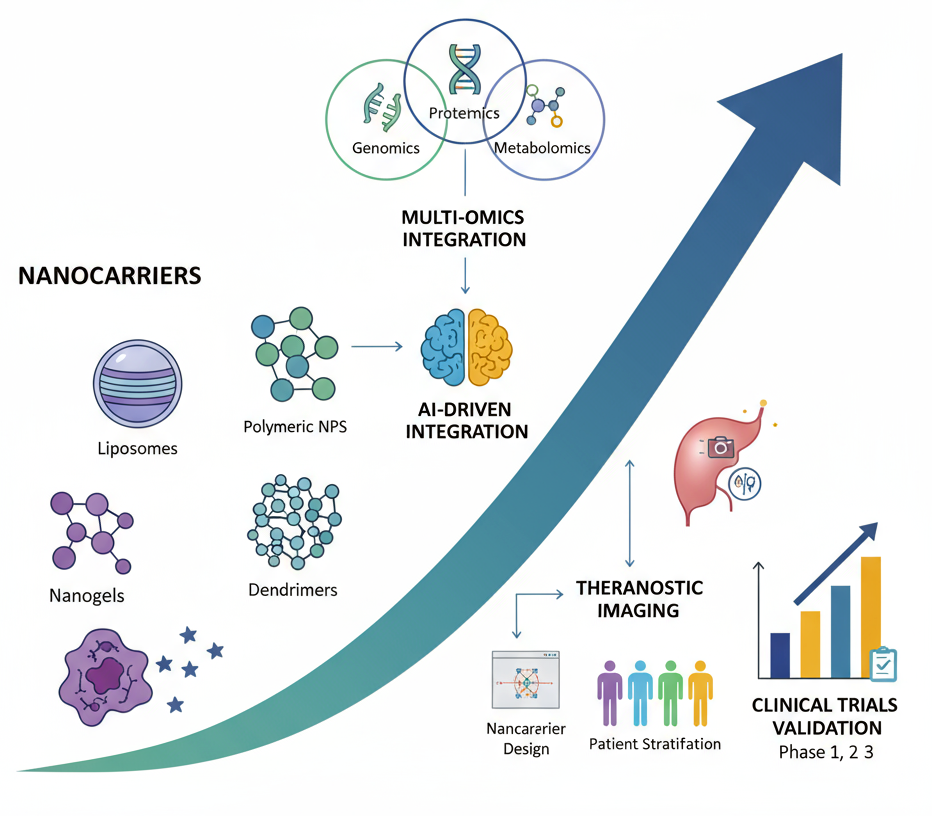
Precision oncology has emerged as a transformative paradigm, tailoring cancer therapies to the molecular and clinical profiles of individual patients. Nanomedicine plays a central role in this shift, offering targeted drug delivery, improved bioavailability, reduced systemic toxicity, and theranostic capabilities that combine therapy and diagnostics. This systematic review evaluates recent advances in nanomedicine for precision oncology from 2019 to 2024, emphasizing multi-cancer applications, clinical trial outcomes, and the integration of artificial intelligence (AI) and omics-based strategies. Relevant studies and trials were retrieved from PubMed, Scopus, and ClinicalTrials.gov following PRISMA guidelines, focusing on nanomedicine interventions, clinical validation, and AI-omics integration. Progress has been documented across diverse malignancies, including breast, lung, colorectal, prostate, hematological, pediatric, and refractory cancers. Clinical trial evidence highlights the superior safety and therapeutic indices of liposomes, polymeric nanoparticles, dendrimers, and nanogels compared with conventional treatments. AI-driven tools have accelerated nanocarrier design, enhanced imaging precision, optimized patient stratification, and improved real-time treatment monitoring. Omics approaches genomics, proteomics, and metabolomics have enriched tumor biology insights, enabling highly personalized nanomedicine formulations. However, key challenges remain, such as tumor microenvironment barriers, immunogenicity, long-term safety, regulatory uncertainties, and limited large-scale validation. The convergence of nanomedicine, AI, and omics holds immense promise for dynamic, patient-specific cancer therapy.
Downloads
Published
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.





